Biodegradable shale inhibitor developed for water-based muds
Salt-free chemistries ID’d for drilling in sensitive, highly regulated environments
By Andy Bradbury, Justin Mclellan, Paul Craddock, Jane Arthur, Impact Fluid Solutions; Michael Poindexter, Cole Witham, Cecile Boyer, The Dow Chemical Co

Global developments in the shale inhibition market are trending toward environmentally friendly materials that are biodegradable, non-bioaccumulative, non-toxic and ultimately non-hazardous. Further, many markets are moving toward or already mandate salt-free systems due to environmental regulations, disposal needs and potential groundwater contamination. This article will detail efforts to develop new shale inhibitors that satisfy these requirements while maintaining performance.
Through a structured research program, groups of novel candidate chemistries in several homologous series were studied and validated using shale recovery, hardness and slake durability (roller cage) tests. This program identified inhibitors with superior performance across multiple parameters, both technical and environmental. Additionally, all identified new materials having excellent performance did not require the addition of a counter-ion, e.g., potassium chloride salt. Results will be presented as a study into the effects that sequential molecular structure changes have on shale inhibitor performance. The chemistries that were identified from the study exhibited improved inhibitor performance relative to known glycol/salt materials and other commonly used amines.
Background
Shales account for more than 75% of all drilled formations, and more than 70% of borehole problems relate to shale instability. Shales are also reported to cause more problems per foot drilled than any other type of formation. Over the past 10 years, reactive shale ranked as one of the top ten mud-related issues affecting deepwater drilling operations. Reactive shale is also a major contributor to stuck pipe, another concern in the top 10 list. These statements should be considered with respect to the drilling fluid used and the type of shale that is encountered. It is the interaction of these two factors that largely determines the extent of the problem.
The reactivity of shale toward water is a much studied and debated topic. For years, the swelling of shale, which is due to the ingress of water and hydration of metal cations, has been considered the “best documented” and main failure mechanism. Combinations of swelling, dispersion, migration and swelling-induced migration have all been given extensive attention. Water activity, brine composition, brine concentration and pH have all been shown to play a role in shale stability. If shale instability is not properly addressed, then the consequences can be significant, including complete loss of the well.
This article will report on the development of shale inhibitors that increase the performance of water-based muds (WBMs). The service industry has devoted a significant amount of effort to bring the performance of WBMs up to that of oil-based muds (OBMs). To abate the unwanted reactions of reactive shales with water, WBMs contain shale inhibitors that range from salts to simple glycols that often contain salts to various amines to polymers. Collaborative work between industry and universities has put forth several mechanisms for the polyetheramine class of shale inhibitors which is considered among the best-in-class for low molecular weight inhibitors. More such studies are needed to progress understanding of shale inhibition.

One approach to study how molecular structure influences shale inhibition performance is to pick a particular molecular descriptor or series of descriptors (e.g., molecular weight, degree of branching, varying heteroatom content, length of hydrophilic/hydrophobic segments, number of end groups bearing a particular functionality, etc), then make different homologous series where the descriptors are systematically varied and conduct comparative tests. Most comparative testing for this work used shale recovery and hardness performance parameters as a means to differentiate the various candidates. While such studies generally cannot provide conclusive evidence regarding the mechanism of inhibition, they are highly informative regarding critical molecular features needed for performance. Such features can suggest reasonable assumptions regarding mode of action. The first level of screening for all materials was recovery and hardness performance on London shale samples. If a given material demonstrated outstanding inhibitory performance on this swelling shale, it was subjected to broader and more demanding performance tests on London and oxford shale, and analyzed for biodegradability and toxicity, among other environmental factors.
Test methodology
When attempting an in-depth study of the effect of subtle changes that chemical structure can have on shale inhibition, the key step is to secure a large enough volume of a consistent shale material to test against. The mineralogical makeup and physiochemical nature of shales are important criteria in how they will behave.
Due to the size of our study, outcrop shales of known, characterized mineralogy were used. The advantage to such substrates is the availability of large shale quantities with similar composition and properties. However, there is a caveat for choosing outcrop shale, which is that the chemical makeup of such shales is one of higher water activity and structurally of poorer consolidation than shales that are drilled at depth. To compensate, a range of different outcrop samples were used that span the shale types that could be encountered while drilling; these contain clays from highly swelling through mixed clays to highly dispersive. It’s also acknowledged that the laboratory testing used for this testing is probably more onerous for shale inhibition performance than that seen in the field because of the above factors.
The outcrop shales used for this study were London Shale and Oxford Shale. The former is a marine geological formation from the Lower Eocene period and is a reasonable analogue of Tertiary shale found in upper hole sections and typically contains 15-25% smectite clay. The shale is plastic in nature due to its moisture content and has low bulk density. This shale is recognized by operators and mud companies as a representative swelling shale.
The Oxford shale formation is a Jurassic marine sedimentary rock formation, which is less swelling than London shale as it contains less smectite but more illite clay. It is dispersible and effective for hardness testing. It is also recognized as an acceptable analogue of problems encountered during drilling and has a mixture of swelling and dispersive character.
Shale testing
The procedures outlined below were used to evaluate the inhibiting chemistry homologous series and benchmark relative performance. The standard test protocol includes:
• Bottle recovery tests;
• Slake durability (roller cage) recovery tests (more aggressive test);
• Shale hardness test (sometimes referred to as the BP hardness tester); and
• Shale moisture content change.
For all tests, it’s recognized that the rheological properties of the fluid can influence the test results by altering the amount of agitation in the rolling phase. For these tests, the rheological parameters of each fluid tested are designed to be similar to minimize any inaccuracies in cross-fluid comparisons.
In essence, the shale performance efficiency of each chemistry was tested on an eight-point matrix, and enhanced performance was sought across all data points. This then leads into toxicity and biodegradation characterization against the same molecular changes within each homologous series.
Results and discussion
Series 1 (quaternary amine-containing molecules)
To better understand the structural properties that lead to successful shale inhibition performance, a series of hydrophobically modified, quaternary amine containing molecules was synthesized and analyzed for shale recovery and hardness. By first analyzing for the relative inhibitory performance of the materials, higher-performing materials could be readily identified and promoted for further inhibition and environmental testing. Using London shale, an optimal length of the hydrophobic moiety was determined for best recovery and hardness. Since the size of the molecule is dictated by its hydrophobic functionality, an optimal length corresponds to an optimal molecular weight.
Additionally, a series of quaternary amine-containing molecules were synthesized with a high charge density and increasing amounts of hydrophilicity. For this series, it was found that increasing either the molecular weight or the hydrophilicity improves the recovery and hardness as tested with London shale. Overall, results indicate that new solutions to the shale swelling and dispersion problem can be found with new chemistry.
Series 2 (substituted amines with alcohol functionality)
While new quaternary amine-containing molecules can improve the shale inhibition performance of WBMs, it is preferable to use ion/salt-free materials. To this end, substituted amines having alcohol functionalities provided different chemical classes and were synthesized with varying molecular weight, amine content and branching. Similar to the studies based on quaternary amine-containing molecules, it was found that the best shale inhibition performance could be obtained when an optimal molecular weight and amine content were used in combination with increased branching.
The best molecule from this series was identified by analysis of the shale recovery and hardness data (Chemistry A). It was then subjected to further investigations and demonstrated excellent performance relative to industry standards.
Interestingly, related materials that have the same functionalities (i.e., amines and alcohols) but different structures do not perform well, indicating that only particular substituted amines with alcohols as specific groupings generate enhanced inhibition performance. While trends within a series of related structures are clear, understanding the effects when expanding hypotheses to related materials remains an active area of investigation.
Series 3 (primary amines with hydrophobic groups – no alcohols)
A series of primary amines with no alcohol functionality but with hydrophobic groups was also found to demonstrate excellent performance having optimal structures. As with other materials studied, a balance exists between molecular weight and hydrophobic length that generates peak shale inhibition. The recovery of shale is also improved by increasing the hydrophobicity of the molecule, by increasing branching or by using an optimal molecular weight with high hydrophobicity. However, hardness of the cuttings will increase with decreasing molecular weight, similar to recovery, but branching and hydrophobicity have a minimal effect.
The best-in-class performer from this study was Chemistry B.
Chemistries A and B
Two chemistries came to light in this investigation that had good properties across the shale substrate testing. Chemistry A is a commercially available material that produces very hard cuttings and good shale inhibition characteristics at concentrations of 2% to 3% v/v. Its performance can be compared against industry-standard amines and glycol in freshwater. Chemistry B is a developmental material that has either equivalent or, in some tests, incrementally better shale characteristics over a broader concentration range of 1% to 3% but is also readily biodegradable, which advances its environmental credentials.
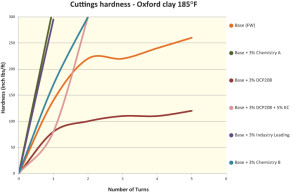
The evaluation of these two shale inhibitor chemistries, both in terms of hardness and slake durability, is shown in Figures 1 and 2 on London clay, which is the more swelling version. Figure 1 illustrates how Chemistries A and B exhibit improved hardness performance over that of an industry-leading amine, reaching higher torque with less turns at the same dosage.
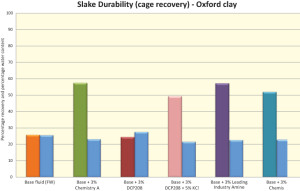
It is important to note that the fluids for Chemistries A and B are freshwater systems. The standard glycol exhibited lower hardness than Chemistries A and B and the industry-leading amine, and the former required KCl as an adjunct to perform. This feature defeated the objective of having a chloride-free or freshwater drilling fluid. The slake durability performance of Chemistry B stands out above that of other materials tested in this study (Figure 2).
The same chemistries were evaluated for hardness and slake durability on Oxford shale. Results are shown in Figures 3 and 4. Chemistry A, B and the industry-leading amine top the hardness performance data as they develop higher torques faster than the other tested materials (Figure 3). The slake durability performance of Chemistry A, B and the industry-leading amine are comparable in this study, with Chemistry A showing a slight advantage (Figure 4).
Second-gen inhibitor
For situations where higher performance is required and greater environmental constraints are present, a second-generation inhibitor for WBMs is being developed.The experimental methodology successfully identified Chemistry A as a top shale inhibitor. These experiments also indicated another top performer in Chemistry B. Initial results indicate that the new material has inhibition performance exceeding that of leading industry materials that do not require the addition of salts. Both recovery and hardness tests suggest high efficacy at comparable dosage levels (Figures 1 and 3) and initial slake durability studies are also encouraging (Figures 2 and 4).
Beyond its performance attributes, several tests suggest a superior environmental profile. EPA’s EPI Suite (BIOWIN models) estimated that this inhibitor would be readily biodegradable under aerobic conditions. This prediction was confirmed experimentally via a screening test based on the OECD 301F guideline. According to this test, the inhibitor can be considered “readily biodegradable” because it reached 120% biodegradation at the end of the 28-day study. The 120% is based on biological oxygen demand at 28 days compared with the theoretical oxygen demand of the test material. The biodegradation percentage in this OECD 301F test may be higher than 100% due to additional oxygen consumption other than the amount required for the bio-oxidation of the test material. Dissolved organic carbon removal was 98% after 28 days, which suggests that the test material was completely degraded. These results are being validated by a more stringent GLP OECD 306 biodegradation test in seawater.
A quantitative structure activity relationship (QSAR) assessment of aquatic toxicity of the inhibitor, in pure form, was performed using the OECD QSAR toolbox and a relevant subset of model molecules from databases of aquatic toxicity results. According to the US EPA classification, the derived model predicted the pure compound to be moderately toxic to algae, slightly toxic to Daphnia Magna and practically non-toxic to fish. Practically speaking, aquatic species do not have a high potential for exposure to the inhibitor in pure form but would be more prone to encounter drilling fluid formulations. Thus, testing for the toxicity of fully formulated drilling fluids toward aquatic species is more relevant and would often be expected to be less than that of the pure compound for each respective species, as predicted above.
A 96-hr definitive bioassay was conducted on a Generic 7 drilling fluid containing up to 2 wt% of the inhibitor following EPA’s drilling fluid toxicity test guidelines. The results showed a LC50 value, the concentration at which 50% of the test organisms respond, above 50,000 ppm, thus passing the guideline minimum requirement of 30,000 ppm. This indicates that the second-generation inhibitor may generate remarkable inhibition performance, as well as have minimal environmental impact.
Environmental data for Chemistries A, B and an industry-leading amine that encompass toxicity and biodegradation test results, as well as partition coefficients, shows an evolution in improvements in toxicity and biodegradation results within the series. Chemistry A exhibits good toxicity data but poor biodegradation, an element that Chemistry B improves upon. A high-end industry-leading amine, despite an excellent shale performance, can have very poor toxicity and biodegradation characteristics.
Conclusions
The main goal for this investigation was to identify new chemistries that have enhanced shale inhibition characteristics that could be in used in freshwater systems and which also have enhanced toxicity and biodegradation profiles. The focus of these efforts would be for land drilling in environmentally sensitive locations, where environmental regulations are either stringent or are becoming more challenging.
Extensive experiments were performed to identify not only new chemistries but also structural factors that contribute to excellent shale inhibition. A novel chemistry was identified and developed that shows excellent performance across economic, technical and environmental parameters. This chemistry represents an excellent replacement for glycol/KCl systems. A next-generation inhibitor with superior performance for demanding applications was also identified and is being validated.
The methodology presented allowed the development of new chemistry and an improved understanding of how molecular structure affects both shale inhibition performance and environmental impact. This knowledge will enable continued product development and will provide the drilling industry with improved materials to mitigate formation damage.
This article is based on a presentation at the 2013 AADE National Technical Conference & Exhibition, Oklahoma City, Okla., 26-27 February.
References
1. Lal, M. Shale Stability: Drilling Fluid Interaction and Shale Strength. SPE Paper 54356; Presented at the Latin American and Caribbean Petroleum Engineering Conference, Caracas, 21-21 April 1999.
2. Bloy, B.; Davis, N.; Smolen, B.; Bailey, L.; Houwen, O.; Reid, P.; Sherwood, J. Fraser, L.; Hodder, M. Designing and Managing Drilling Fluid. Oilfield Rev. 1994, April, 33-43.
3. McLean, A.; Wilde, A.; Zamora, M.; Rafferty, M. “The Top 10 Mud-Related Concerns in Deepwater Drilling Operations – Revisited After 10 Years,” Paper AADE-10-DF-HO-04; Presented at the American Association of Drilling Engineers Conference, Houston, 6-7 April 2010.
4. Zamora, M.; Broussard, P. N.; Stephens, M.P. “The Top 10 Mud-Related Concerns in Deepwater Drilling Operations,” SPE Paper 59019; Presented at the International Petroleum Conference and Exhibition, Villahermosa, 1-3 February 2000.
5. Santarelli, F. J.; Carminati, S. Do Shales Swell? A Critical Review of Available Evidence. Paper 29421; Presented at the SPE/IADC Drilling Conference, Amsterdam, 28 February – 2 March 1995.
6. Gray, D. H.; Rex, R. W. Formation Damage in Sandstones Caused by Clay Dispersion and Migration. Clays & Clay Minerals 1966, 14, 355-366.
7. Reid, P. I.; Harrington, P. M.; Minton, R. C. Shale Tests Help Develop Inhibitive Water-Based Muds. Ocean Ind. 1991, October, 19-24.
8. Mohan, K. K.; Vaidya, R. N.; Reed, M. G.; Fogler, H. S. Water Sensitivity of Sandstones Containing Swelling and Non-Swelling Clays. Coll. Surf. A: Physico & Eng. Aspects 1993, 73, 237-254.
9. van Oort, E. On the Physical and Chemical Stability of Shales. J. Petr. Sci. Eng. 2003, 38, 213-235.
10. Sawdon, C. A.; Brady, M. E.; Cliffe, S.; James, S. G. An Integrated Approach to the Development of a Water-Based Mud with Oil-Based Mud Performance. Paper 29375; Presented at the SPE/IADC Drilling Conference, Amsterdam, 28 February – 2 March 1995.
11. Hoover, E.; Trenery, J.; Mullen, G.; Benaissa, S.; Richards, L. “New Water-Based Fluid for Increased Onshore Drilling Performance,” Paper AADE-08-DF-HO-38; Presented at the American Association of Drilling Engineers Conference, Houston, 8-9 April 2008.
12. Boek, E. S.; Coveney, P. V.; Skipper, N. T. Monte Carlo Molecular Modeling Studies of Hydrated Li-, Na-, and K-Smectites: Understanding the Role of Potassium as a Clay Swelling Inhibitor. J. Am Chem. Soc. 1995, 117, 12608-12617.
13. Reid, P. I.; Elliot, G. P.; Minton, R. C.; Chambers, B. D.; Burt, D. A. Reduced Environmental Impact and Improved Drilling Performance with Water-Based Muds Containing Glycols. Paper 25989; Presented at the SPE/EPA Exploration & Production Environmental Conference, San Antonio, 7-10 March 1993.
14. Aston, M. S.; Elliot, G. P. Water-Based Glycol Drilling Muds: Shale Inhibition Mechanisms. Paper 28818; Presented at the SPE European Petroleum Conference, London, 25-27 October 1994.
15. Reid, P. I.; Dolan, B.; Cliffe, S. Mechanism of Shale Inhibition by Polyols in Water Based Drilling Fluids. Paper 28960; Presented at the SPE International Symposium on Oilfield Chemistry, San Antonio, 14-17 February 1995.
16. Patel, A. D. Design and Development of Quaternary Amine Compounds: Shale Inhibition with Improved Environmental Profile. Paper 121737; Presented at the SPE International Symposium on Oilfield Chemistry, The Woodlands, 20-22 April 2009.
17. Young, S.; Stamatakis, E. Novel Inhibitor Chemistry Stabilizes Shales. Paper #6; Presented at the American Association of Drilling Engineers Technical Conference, Houston, 11-12 April 2006.
18. Zhang, L.-M.; Sun, B.-W. Inhibition of Water-Soluble Cationic Cellulosic Polymers to Clay Hydration. J. Appl. Polymer Sci. 2002, 74, 3008-3093.
19. Bruton, J. R.; McLaurine, H. C. Modified Poly-Amino Acid Hydration Suppressant Proves Successful in Controlling Reactive Shales. Paper 26327; Presented at the 68th Annual SPE Technical Conference and Exhibition, Houston, 3-6 October 1993.
20. Hodder, M.; Cliffe, S.; Greenwell, C.; Williams, P. Coveney, P. Clay Swelling Inhibitors – Computer Design and Validation. Paper #32; Presented at the American Association of Drilling Engineers Technical Conference, Houston, 6-7 April 2010.
21. Patel, A. D.; Stamatakis, E.; Davis, E. Shale Hydration Inhibition Agent and Method of Use. U.S. Patent 6,609,578; issued August 26, 2003.
22. Young S, M-I Swaco, Friedheim, J. Testing protocols for optimized drilling fluid design. This paper was presented at the Offshore Mediterranean Conference and Exhibition in Ravenna, Italy, March 25-27, 2009
23. OECD 301F Guideline for testing of chemicals, Ready biodegradability. Manometric respirometry, July 17, 1992.
24. Drilling Fluid Toxicity Test, Federal Register, Vol. 50, No. 165, Aug 26, 1985, pp 34631-34636.